Tiny Drifters, Big Impact: Zooplankton and Biodiversity
By Joshua Matthews
Note: This blog was originally created for a university class blog project in the fall semester of 2024. It is based on a Literature Review paper for the same class instructed by Dr. Sabrina Russo. You can find my full Literature Review paper on the environmental factors affecting zooplankton equilibrium dynamics here.
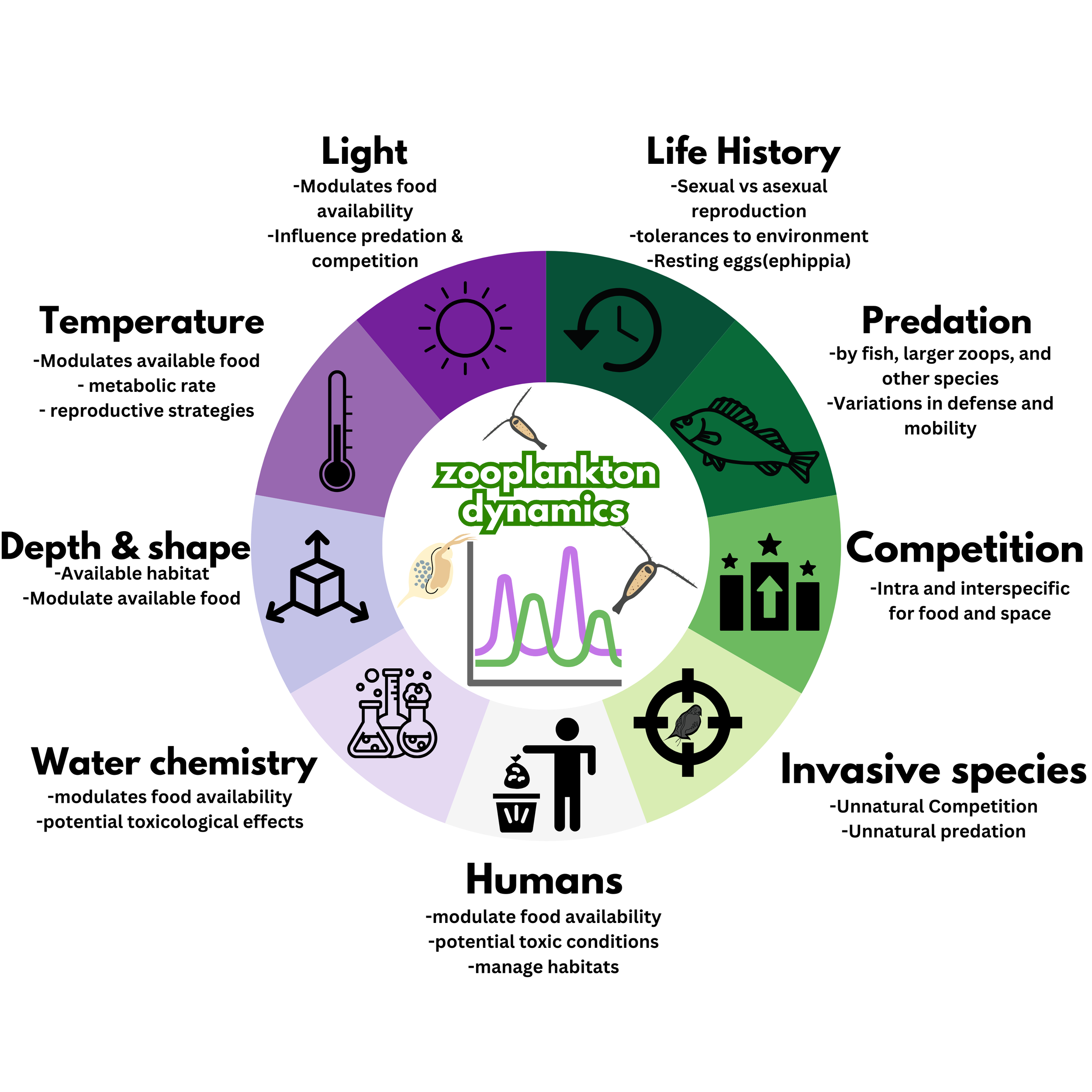
Zooplankton are tiny drifters in nearly all freshwater and marine ecosystems and act as regulators of other microorganisms as well as support larger creatures such as fish (Brooks 1968). There are thousands of species of zooplankters making them a diverse subset of creatures. By grazing on algae and cycling nutrients zooplankton support a microorganism diversity in aquatic ecosystems (Brooks 1968). They also do this by acting as feed for larger creatures including many fish who rely on them as an important source of food during their young developmental stages. Healthy zooplankton populations support healthy aquatic ecosystems. While they play several important ecological roles, zooplankton are also sensitive to the dynamic conditions within the body of water they live in (Sommer and Stibor 2002). Their populations and communities are affected by factors including predation, competition, invasive species, available light, water temperature and chemical composition, waterbody shape and depth (Figure 1). Humans have the ability to alter these influencing factors thus shaping the biodiversity of earths waters.
These tiny creatures drift through various inland bodies of water, including lakes, ponds, reservoirs, and wetlands (collectively referred to as lakes). They are of ecological importance due to performing top-down structuring of aquatic food webs by grazing on microorganisms and bottom-up structuring of larger species that feed on zooplankton (Brooks 1968). Zooplankters perform top-down structuring of aquatic ecosystems by grazing on algae, bacteria, and other microparticles that fit into their mouths. They perform bottom-up structuring of food webs as a source of feed to larger creatures, especially small fish. A lack of zooplankton when fish hatch can lead to poor survivability among fish fry. This top-down and bottom-up structuring of aquatic ecosystems by zooplankton make them key players in maintaining biodiversity. Through this blog we will explore how various environmental conditions and ecological interactions affects biodiversity among zooplankton.
Zooplankton come in many shapes and sizes and are a diverse group of creatures with thousands of species. Here we will explore how two different groups of zooplankton respond to environmental conditions: Cladocera and Copepods. Cladocera include the Daphniidae family, which are commonly known as water flees and look like small peep shaped blobs with a single eye and two branching arms coming off their upper body. Copepods include the Cyclopoid and Calanoid families which resemble little shrimp with a cyclops eye. Zooplankton can quickly respond to the dynamic conditions of the lakes they reside in as they have short life cycles (Sommer and Stibor 2002). Due to their varying uses of and responses to changing environmental conditions, zooplankton biodiversity in a lake can appear to change throughout a lake over the course of a year.
Image 1
On the left is a cyclopoid member of the Copepod group. On the right is a daphnia member of the Cladocera group. Image source: Josh Matthews
As demonstrated in Figure 1, many factors influence zooplankton biodiversity, and humans have varying degrees of control over these factors. Some of these factors directly impact zooplankton, such as predation or responses to changing environmental conditions. Other factors impact the quality and quantity of food resources such as competition, the nutrient content of the lake, available light, and temperature. Zooplankton species have different feeding strategies and tolerances for dynamic environmental conditions (Sommer and Sommer 2006).
Invasive species play a huge role in shaping zooplankton community composition and biodiversity. This mainly happens by increasing pressures from competition and predation which are demonstrated in Figure 1. Invasive species can outcompete zooplankton and other native species for food by being faster and more efficient filter feeders. By quickly consuming algae and other small, suspended particles, invasive species such as zebra and quagga mussels can reduce and, in some cases, even eliminate the food available for zooplankton. Zooplankton are also influenced by predation from fish and other creatures that consume them. Invasive species can increase predation pressures on zooplankton and in some cases may shift biodiversity by preferentially preying on specific species of zooplankton.
Figure 1. Factors that affect zooplankton community biodiversity
Each factor has been assigned a segment along the circle, representing the interconnectedness of all factors. Segments in shades of green represent interactions with other living creatures that affect zooplankton populations and communities, while segments in shades of purple represent interactions with the environment that affect zooplankton communities with human impacts represented in a light gray at the bottom. Segments closer to humans in lighter shades of their respective colors represent the factors that are most easily influenced by humans while factors with darker shades are more difficult for humans to influence.
Zooplankton can consume many different algae particles. Cladocera are filter feeders and will eat whatever fits into their mouths, while copepods tend to be a bit more selective in what they consume (Sommer and Stibor 2002). In general, Cladocera consume smaller algae particles while copepods consume larger particles (Sommer and Sommer 2006). The algae they consume is heavily influenced by the physical and chemical properties of the body of water they live in. Algae is a diverse group of microscopic photosynthesizers that come in many shapes and sizes, each responding differently to environmental conditions. The shifts in algae community composition can also result in shifts in zooplankton community composition as the quality and quantity of available food changes. Algae require moderate temperatures, sunlight, carbon dioxide, water, and nutrients to grow. As the seasonal availability of these resources change, so too does the quality and quantity of food available to zooplankton communities.
The physical and chemical properties of a body of water can also have direct impacts on the zooplankton themselves. Cladocera tend to reproduce via a process known as cyclical parthenogenesis where under ideal conditions females asexually reproduce clones (Decaestecker 2009). Descaestecker suggests that as environmental conditions become unfavorable, such as extreme high or low temperatures and reduction of food resources. They produce males who then sexually mate with the female clones to produce a resting egg called and ephippia. These resting eggs can withstand long periods of unfavorable conditions, even completely drying out. Copepods always reproduce sexually requiring both the male and female to fertilize and produce eggs (Marcus 1982). Unlike the Cladocera which produce babies that closely resemble a full-grown adult, Copepod eggs hatch into a form known as a nauplii which undergoes several transformations while developing into an adult (Sommer and Stibor 2002). Copepods are generally more tolerant to unfavorable environmental conditions but are also capable of producing a resting egg which increases the chances of population rebound after extreme weather conditions such as freezing or drying out of a body of water. Due to their simpler life cycle, Cladocera tend to reach sexual maturity in just a few days, while Copepods tend to take around a month to reach sexual maturity (Sommer and Stibor 2002). These differences in lifecycle can impact their ability to respond to environmental conditions and support ecosystem biodiversity.
As we have discussed and is highlighted in Figure 1, many of the physical and chemical properties of a body of water can affect zooplankton and the algae they eat such as how much light is available, water temperature, and the nutrient chemical composition of the water. Zooplankton are also affected by the shape and depth of the lake as deeper lakes and those with more complex shapes tend to provide more unique habitats for them which influences both community composition and species diversity (Miyashita et al. 2014).
Humans have an outsized influence on affecting nearly all factors from Figure 1 which influence zooplankton communities and diversity. Humans are the number one transporter of invasive species, both intentionally and by accident (Havel et al. 2015). Human activities such as fertilizing field, gardens, and golf courses, as well as disposing of waste such a sewage can add nutrients to bodies of water resulting in algae blooms which change the quality and quantity of food available to zooplankton (Yu et al. 2020). Humans also introduce other pollutants such as heavy metals into water which can have detrimental effects on zooplankton biodiversity a some species are more capable of tolerating them than others (Gagneten 2011). Bodies of water around the globe are unreasoningly getting warmer because of global warming, which in some cases can cause changes to zooplankton biodiversity. Humans also introduce heat into aquatic ecosystems through the discharge of water used to cool power generating and other industrial machines. These direct inputs of hot water have much more pronounced impacts on zooplankton biodiversity than the relatively show and small increases from global warming (Mulhollem et al. 2016). Humans can even impact the available light to support phytoplankton growth for example by the increasingly popular practice of covering lakes and small bodies of water with floating solar panels to generate electricity. While the impact in large lakes may be near negligible, small lakes that have a greater percentage of their surface covered can experience impacts on the quantity and quality of algae available for zooplankton consumption (Nobre et al. 2022).
In his 1961 paper, Hutchinson introduces the paradox of the plankton, where many species thrive on a seemingly limited set of resources. The differential responses of zooplankton communities to varying environmental conditions may explain how they overcome this paradox by occupying highly specialized ecological niches (Record et al. 2014, Sommer and Stibor 2002). When it comes to the microscopic world, there are many ways for creatures to become specialists and support increased in biodiversity. Due to their ecological importance, it has been suggested that zooplankton can be used as bioindicators of ecosystem health (Serranito et al. 2016, Lomartire et al. 2021). Because of the ecosystem services they provide and their differential response to environmental stressors, studying zooplankton can provide insight to inform conservation and management decision making to support biodiversity in aquatic ecosystems (Johnston et al., 2022).
References Cited
Brooks, J., 1968. Effects of Prey Size Selection by Lake Planktivores. SYSTEMATIC ZOOLOGY 17, 273-+. https://doi.org/10.2307/2412007
Decaestecker, E., De Meester, L., Mergeay, J., 2009. Cyclical Parthenogenesis in Daphnia: Sexual Versus Asexual Reproduction, LOST SEX: THE EVOLUTIONARY BIOLOGY OF PARTHENOGENESIS. Springer, Dordrecht. https://doi.org/10.1007/978-90-481-2770-2_15
Gagneten, A.M., 2011. Effects of Contamination by Heavy Metals and Eutrophication on Zooplankton, and Their Possible Effects on the Trophic Webs of Freshwater Aquatic Ecosystems, in: Ansari, A.A., Singh Gill, S., Lanza, G.R., Rast, W. (Eds.), Eutrophication: Causes, Consequences and Control. Springer Netherlands, Dordrecht, pp. 211–223. https://doi.org/10.1007/978-90-481-9625-8_10
Havel, J.E., Kovalenko, K.E., Thomaz, S.M., Amalfitano, S., Kats, L.B., 2015. Aquatic invasive species: challenges for the future. Hydrobiologia 750, 147–170. https://doi.org/10.1007/s10750-014-2166-0
Hutchinson, G.E., 1961. The Paradox of the Plankton. The American Naturalist 95, 137–145. https://doi.org/10.1086/282171
Johnston, N.M., Murphy, E.J., Atkinson, A., Constable, A.J., Cotté, C., Cox, M., Daly, K.L., Driscoll, R., Flores, H., Halfter, S., Henschke, N., Hill, S.L., Höfer, J., Hunt, B.P.V., Kawaguchi, S., Lindsay, D., Liszka, C., Loeb, V., Manno, C., Meyer, B., Pakhomov, E.A., Pinkerton, M.H., Reiss, C.S., Richerson, K., Jr., W.O.S., Steinberg, D.K., Swadling, K.M., Tarling, G.A., Thorpe, S.E., Veytia, D., Ward, P., Weldrick, C.K., Yang, G., 2022. Status, Change, and Futures of Zooplankton in the Southern Ocean. Front. Ecol. Evol. 9. https://doi.org/10.3389/fevo.2021.624692
Lomartire, S., Marques, J.C., Goncalves, A.M.M., 2021. The key role of zooplankton in ecosystem services: A perspective of interaction between zooplankton and fish recruitment. Ecol. Indic. 129, 107867. https://doi.org/10.1016/j.ecolind.2021.107867
Marcus, N., 1982. Photoperiodic and Temperature Regulation of Diapause in Labidocera-Aestiva (copepoda, Calanoida). Biol. Bull. 162, 45–52. https://doi.org/10.2307/1540969
Miyashita, T., Amano, T., Yamakita, T., 2014. Effects of Ecosystem Diversity on Species Richness and Ecosystem Functioning and Services: A General Conceptualization, in: Nakano, S.I., Yahara, T., Nakashizuka, T. (Eds.), INTEGRATIVE OBSERVATIONS AND ASSESSMENTS, Ecological Research Monographs. Springer-Verlag Berlin, Berlin, pp. 29–47. https://doi.org/10.1007/978-4-431-54783-9_2
Mulhollem, J.J., Colombo, R.E., Wahl, D.H., 2016. Effects of heated effluent on Midwestern US lakes: implications for future climate change. Aquat Sci 78, 743–753. https://doi.org/10.1007/s00027-016-0466-3
Nobre, R., Bouletreau, S., Cucherousset, J., 2022. Small lakes at risk from extensive solar-panel coverage. Nature 607, 239–239.
Record, N.R., Pershing, A.J., Maps, F., 2014. The paradox of the “paradox of the plankton.” ICES J. Mar. Sci. 71, 236–240. https://doi.org/10.1093/icesjms/fst049
Serranito, B., Aubert, A., Stemmann, L., Rossi, N., Jamet, J.L., 2016. Proposition of indicators of anthropogenic pressure in the Bay of Toulon (Mediterranean Sea) based on zooplankton time-series. Cont. Shelf Res. 121, 3–12. https://doi.org/10.1016/j.csr.2016.01.016
Sommer, U., Sommer, F., 2006. Cladocerans versus copepods: the cause of contrasting top-down controls on freshwater and marine phytoplankton. Oecologia 147, 183–194. https://doi.org/10.1007/s00442-005-0320-0
Sommer, U., Stibor, H., 2002. Copepoda-Cladocera-Tunicata: The role of three major mesozooplankton groups in pelagic food webs. Ecol. Res. 17, 161–174. https://doi.org/10.1046/j.1440-1703.2002.00476.x
Yu, C., Li, Z., Xu, Z., Yang, Z., 2020. Lake recovery from eutrophication: Quantitative response of trophic states to anthropogenic influences. Ecol. Eng. 143, 105697. https://doi.org/10.1016/j.ecoleng.2019.105697